Senseo Electricity Basics 2: Generation
This post is part of the Senseo series:
- Senseo Prelude
- Senseo Electricity Basics 1
- Senseo Electricity Basics 2: Generation
- Senseo Electricity Basics 3: Grid to Wall Socket
- Senseo Boiler: Heat and Electricity
- Senseo Boiler: Sensing Temperature
- Senseo Boiler: Sensing Temperature Part 2
- Senseo Boiler: Safety
- Senseo Boiler: Brewing
- What is Plastic?
- PCB - Printed Circuit Boards: Fundamentals 1
- PCB Fundamentals 2: MOSFET Transistors
- PCB Fundamentals 3: CMOS Logic
- PCB Fundamentals 4: Combinational v Sequential Logic
- PCB Fundamentals 5: D-Latch
- PCB Fundamentals 6: Clocks & Flip-Flops
- PCB Microcontroller Subsystems: CPU core
- PCB Microcontroller Subsystems: GPIO
- Senseo GPIO Button Example
- PCB Microcontroller Subsystems: ADC (Conceptual)
- Senseo Interlude: Considering Quality
Hydroelectricity and Magnets
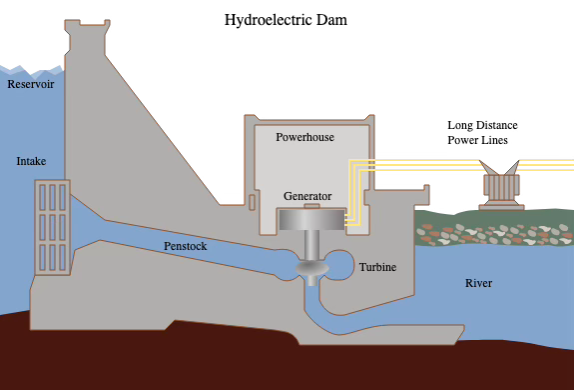
I think the most interesting question that may not be relevant to the Senseo machine is that of how electricity is harnessed from nature. I’d say the easiest way to do this would be to put a damn in a river and go with hydroelectricity. The idea here is that moving water, such has that coming down a dam, creates theoretical energy (kinetic energy) that can be harnessed. So what is done is that you might create a turbine where the water falling down and hits the blades, making it spin. This spinning turbine, being powered by nothing other than gravity, is connected to a generator. A generator is just as we’ve seen before with AC, with a coil of wire (in big loops) and a magnet. That magnet is attached to the rotation that occurs, and a changing magnetic field around the wire is created because of the spinning. And again a law of nature (electromagnetic induction) dictates that changing magnetic fields push electrons inside a wire, making it electricity. The coil in the generator is therefore connected to thick wires leaving the generators, which are in turn connected to the power lines we see in the streets. This same exact idea works also for wind turbines.
Another interesting concept that makes all of this work is the magnet itself. For what really is a magnet? It all starts at the point of atoms, which are made of a nucleus (protons and neutrons) and small electrons spinning around the nucleus. Everything we see is made of atoms. Now as seen before, the electrons are charged and moving and any moving charge creates a magnetic field around it. As such every electron acts like a miniature magnet in the atom. Now in most materials such as wood or copper, electrons are paired up in such a way that their magnetic field points in opposite directions, so when one electron’s field points north and the other’s south, they’ll cancel out with no net magnetism.
Now there are other materials (iron, nickel, cobalt) that have groups of atoms inside (domains) that naturally form and have huge amounts of electrons lined up together in the same direction so as to make magnetic fields. So each domain is it’s own magnet inside the metal. Now quite often in a material like iron, the domains point in random directions, and the metal isn’t magnetic overall as a result. But making magnetism is therefore the same as merely lining up these domains, which happens if we put a separate magnet next to it, the magnetic field makes the domains rotate and align in the same direction, making the iron have a north pole and south pole. So a magnet is physically just a piece of metal in which atomic magnetic fields are lined up. On the one end, the fields will point outwards (north pole) while on the other they’ll point inward (south pole).
Because magnets are in essence just spinning electrons aligned in certain directions, the logic of opposite or like charges attracting and repelling still holds. So when the south pole of one domain is close to the north pole of another, these opposites will attract. Now this might seem confusing since earlier it was said in the hair and comb example that there’s only a magnetic field when there motion in charged particles (=current magnetism), and if that motion stops there remains only an electric field.
There’s a built in exception however inside atoms, where electrons might not be traveling through space, they still spin around the nuclei making each electron behave like a tiny permanent magnet (=permanent magnets). No real magnetic field is created around the object with atoms in it, however, as these small magnetic fields from individual electrons point in random directions and cancel out. So there exist two separate sources of magnetism, one from moving charges, one inside atoms.
Batteries
Aside from generators, which move electrons by moving magnets, another major way in which people have started generating electricity is through batteries. Batteries are essentially moving electrons through chemical energy. As such, a chemical reaction pushes electrons from one side to the other. A battery generally has three parts, a negative side, positive side and a chemical in between. The negative side is full of electrons, while the positive side wants electrons but can only get them from the negative side through an outside wire connecting the two sides. This occurrence is by definition a DC circuit. An empty battery is then nothing more than one with a used up chemical in the middle (or a broken one). Electrons don’t get back to the negative side inside.
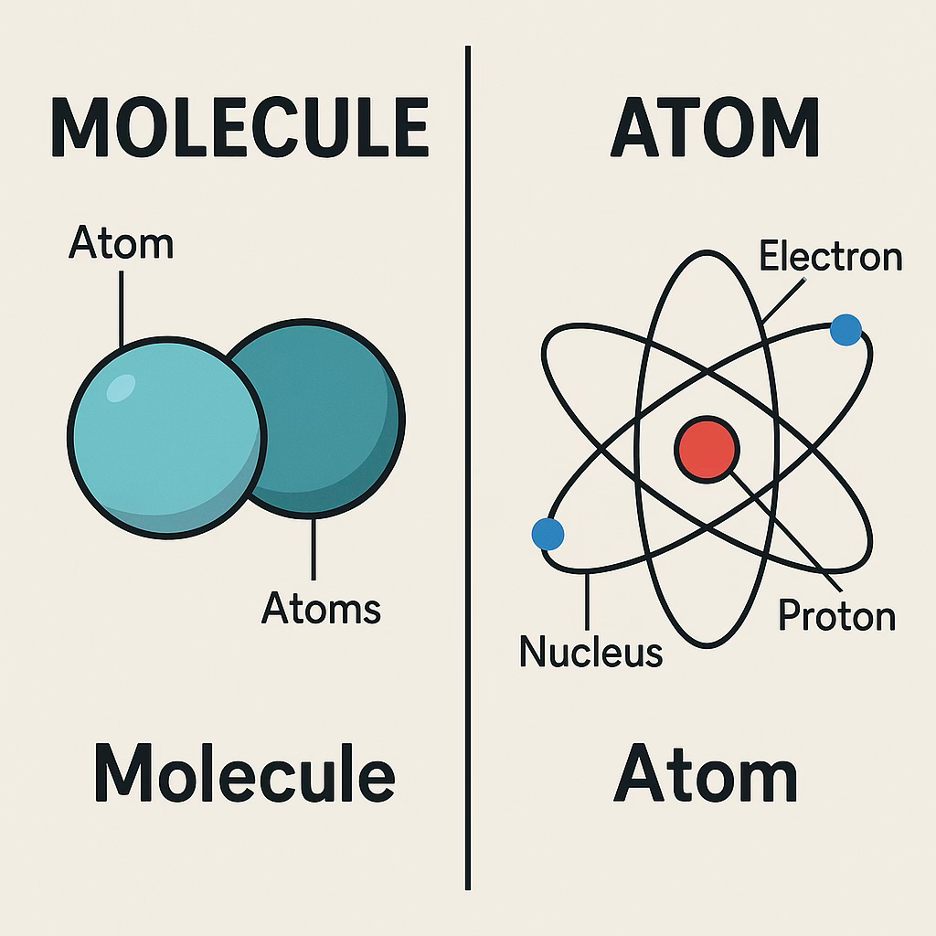
To go a bit deeper into this, we start with going over the basics of chemistry. An atom is the basic building block of everything, like hair or a watch, and is made of protons (+) and neutrons in the core, and electrons moving around it. Molecules, on the other hand, are merely groups of atoms stuck together, such as water, which is 2 hydrogen atoms and an oxygen atom. As such a battery electrolyte (chemical in the middle) is just molecules made from atoms. Next to these, ions are also elementary to the process. an ion is quite easy to understand, an atom has both protons (+) and electrons (-), and equally as much of both so that it’s neutral. Now with an atom type that likes to lose (or gain) its electrons, having actually lost (gained) them makes the charge of the atom not neutral anymore, and such a charged atom is an ion.
Now it is in the nature of some atoms to want to shed their electrons (phenomenon called electronegativity). An atom like zinc (Zn) wants to get rid of its outer electrons, while atoms like copper (Cu) or oxygen (O) really wants extra electrons. So in batteries the negative side has a material like zinc, while the positive holds a material like copper. Because there’s these types of material creating imbalance in electron amounts, voltage is created (how strong electrons are pushed or pulled).
We can perhaps first consider a sitting (copper-zinc) battery with no wire attached to it:
The negative side has zinc metal, which is made of zinc atoms (consisting of a nucleus and electrons orbiting around it). Generally, this side is called the Anode, as it loses electrons. The other electrons are very easily shed for zinc, so it’s eager to create a certain reaction: Zn —> Zn²⁺ + 2 electrons. This reaction entails that the zinc atom becomes a zinc ion which is positive, and two electrons are shed into the metal. This reaction is only done when zinc has somewhere to dump the electrons. Once the battery is built however, the zinc initially does want to start this process. A few zinc atoms at the surface turn into zinc ions Zn²⁺, float into the electrolyte and leave their electrons behind the the zinc metal. The zinc metal therefore becomes a little extra negative (holding extra electrons, which are negative charges). This process stops immediately, however, as now there is a negatively charged zinc metal, and a positively charged liquid (zinc ions are positive), which pull on each other creating an electric field. That electric force prevents the chemical reaction from ensuing (until a wire connects both sides).
This is then what creates the voltage. There is negative charge from electrons piling up in the zinc metal while positive charge is created from zinc ions piling up in the liquid near the zinc. That small electric field resulting from this is what is the voltage (difference in charge between two points).
The positive side has copper metal, which is made of copper atoms that hold onto electrons strongly, so it will never release electrons (Cathode = gains electrons). In the battery, this metal just sits there and only participates when electrons arrive from somewhere. In a sitting battery nothing occurs here. The reason the copper side is considered positive isn’t because of electrons being lost or gained, it’s merely because it’s more positive relative to the zinc side, which has those extra electrons.
electrolyte is the liquid in the middle and is basically copper sulfate (CuSO₄) dissolved into water. When copper sulfate dissolves into water however, it breaks into copper ions Cu²⁺ (positive ions) and sulfate ions SO₄²⁻ (negative ions). These ions therefore just float around in the liquid, not in the metal. They don’t have anything to do with the metals on either side, it just allows ions to move while not allowing electrons to. the chemical barrier acts as a highway for ions but a barrier for electrons.
Now we can consider the battery when a wire attaches both sides:
Anode Zinc used to have electrons built up and zinc ions (Zn²⁺) built up inside while electric forces stopped further reaction. Now, however, the electrons can escape the zinc metal, flow through the wire towards the copper and the zinc side becomes less negative. As there is now a place for electrons to flow, the zinc atoms start dissolving continuously, so that the ion goes into the electrolyte, while the 2 electrons go into the wire. As a result, the zinc metal will slowly get thinner (and get used up).
Cathode Copper suddenly gets electrons, so becomes less negative (relative to the zinc side). Now the electrolyte right next to the copper plates holds these copper ions (Cu²⁺, inherent in the electrolyte) that now see their chance to grab electrons (Cu²⁺ + 2e⁻ → Cu (solid)). By having this reaction, the incoming electrons are turned into a neutral copper atom that sticks onto the copper metal. So the copper metal grows with new metal.
Electrolyte now becomes very active. There are zinc ions (Zn²⁺) accumulating near the zinc metal, copper ions (Cu²⁺) disappearing from near the copper (getting essentially used up over time), and sulfate ions (SO₄²⁻) drifting around balancing the charge. All of these are constantly moving, otherwise the battery stops. Now the circuit must stay balanced everywhere, so when the copper ions disappear at the copper plate, a positive charge is removed from the solution. If nothing were to move to replace it, the liquid near the copper plate would become too negative and the reactions stops. As a result, when the copper ions disappears, the negative sulfate ions (SO₄²⁻) will move elsewhere in the liquid, while the positive zinc ions (Zn²⁺) move towards the copper side to keep the liquid neutral. This same thing occurs at the zinc metal side, where a lot of zinc ions are piling up from the zinc atom reaction, so sulfate ions will move towards the zinc to neutralize this area. So electrons move through the wire creating electricity, while ions move in the liquid (internal current) to keep the battery going.
This summarizes the various ways a battery gets used up: copper ions from the electrolyte get used up from becoming copper atoms, so that no more electrons can get taken in. Zinc atoms get used up by having the dissolving reaction (ion + electrons), so that the zinc metal eventually disappears. At some point also the electrolyte gets saturated because the sulfate ions are outnumbered by zinc ions, making it impossible for the liquid to remain neutral.
Rechargeable batteries
Those reactions causing the battery to die are never going to be undone unfortunately. What a rechargeable battery then does, is choosing materials that do allow for the chemistry to be reversed (batteries are made of loads of materials other than zinc and copper). In such batteries, nothing dissolves permanently into the electrolyte, no atoms get used up (such as zinc), and ions are moving back and forth instead of being destroyed and the structure of the electrodes (the metals) remains intact. What’s essentially being done is that ions are being stored inside the electrode during the design, so no ions are being created or destroyed in the battery. The ions then just simply move back and forth between electrodes (Anode and Cathode).

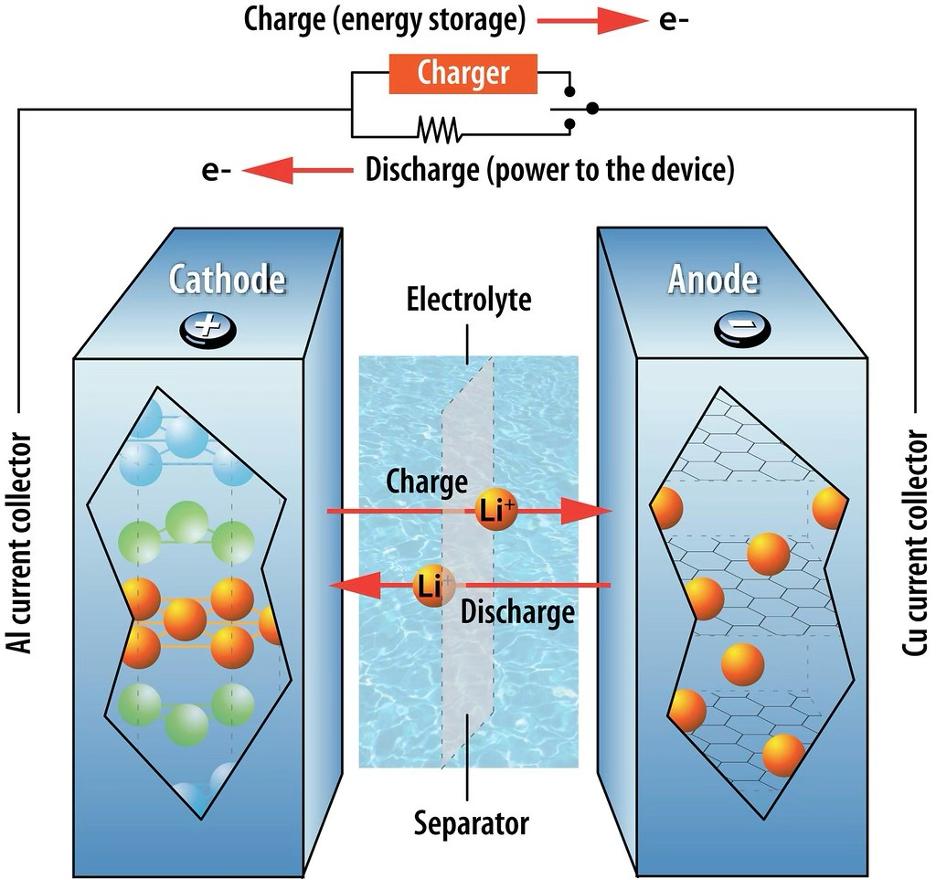
This can best be explained at the hand of the most common rechargeable battery we use today, which is lithium ion (our phones). Such a battery has graphite (layers of carbon) as Anode, lithium metal oxide as Cathode, and lithium salt in organic solvent as electrolyte. So quite a different chemistry than the zinc copper battery we saw earlier. The ions that are stored and moving between sides is then lithium ion (Li⁺). Now the main point with this type of battery is that the metals don’t dissolve and the electrolyte isn’t used up, the lithium ions are just moving between them. While using a device, the battery has electrons going through the wire and ions going through the electrolyte, the ions moving out of the carbon layers (C–Li→C+Li⁺+e⁻) where only carbon stays behind, and into the lithium metal oxide (LiCoO₂+Li⁺+e⁻→Li₂CoO₂) which just absorbs it without changing structure.
Now when we charge the battery, the electrons are kind of forced backwards through the circuit so that electrons go back to the anode, and the lithium ions (Li⁺) go back to the anode. These types of batteries do eventually die, as electrodes (metals) start getting damaged or the ions don’t move back anymore, or if the electrolyte itself just starts degrading in quality.
So the story here is actually very similar to the zinc copper story from before. One the one side we have a stable carbon called graphite, that has sheets and between those are lithium atoms (C-Li can be seen as ‘graphite holding lithium’), the material that likes to lose its electrons. So it’s the lithium atom that loses it’s electron (which goes through the device) and becomes a lithium ion, that moves through the electrolyte into the other side. The graphite therefore becomes the storage place for electrons when the battery discharges (is being used), so that it holds a lot more electrons than the other side, thereby making it negatively charged. Because of this voltage being created as a result of electron difference, the electrons are pushed through the wire. Same idea as before. Big difference, however, is that the ion doesn’t remain in the electrolyte, but instead goes into the Cathode crystal (lithium metal oxide is like a crystal with holes in it).
At the Cathode side, whenever the electrons arrive through the wire, they recombine with the ions that travelled through the electrolyte and sit as atoms inside the metal (CoO₂+Li⁺+e⁻→LiCoO₂). So it’s the lithium atom that is stored again. As such there is no pile up of electrons, keeping the circuit stable. When the battery needs recharging, what has actually occurred is that all lithium atoms have moved from anode to cathode, so that anode has very little lithium left, has no extra electrons as a result and becomes neutral (so no negative charge). Meanwhile at the Cathode side is now full of stored lithium atoms (ions+electrons), so it becomes less positive. The lack of charge imbalance between the sides means no more voltage exists and no electrons are pushed through the wire.
It can be noted here that in the battery design, anode is always made bigger than the cathode, according to the N/P ratio (negative to positive electrode capacity ratio). For instance, if we have a cathode that can store 100 units of lithium, the anode will be made so it can store 110-120. This is done for safety, since if graphite is truly 100% chemically full, the lithium has no place to go anymore, and it will deposit on the surface of the anode instead of inserting into the graphite. So not as lithium ions but as lithium metal. Such raw lithium is flammable and grows spikes (dendrites), such that if a spike grows towards the cathode and touches it, there’s an internal short circuit and battery fires (electrons bypass outside wire, current dumps, battery heats up and thermal runaway occurs —> in short, battery explodes). This story also implies that when our battery is either ‘dead’ or 100% according to out phone, chemically it’s more like 15% or 85%.
Now charging is a pretty interesting process as this links directly to my earlier exploration. When I plug my phone into the wall, the charger first needs to convert the AC current to DC for the charging to work (since batteries also talk in DC). So inside this charger is a transformer, a rectifier, a capacitor and a voltage regulator. This outputs 5V DC for my USB charger, and 9-12V DC for fast charging. By design, the charger will apply a higher voltage than the battery, so for instance 5V, while the battery has 3.7V (or 3V when dead). The charger has two sides, positive and negative, and connects these sides to the positive and negative sides of the battery, respectively. Now as seen before, voltage is electric pressure, how badly electrons want to move, and when the charger from the wall has 5V and the empty battery 3V, it means the charger has a higher electric level. Electrons always flow from lower electric pressure to higher electric pressure because it wants to equalize potential. So when the battery’s anode (graphite) has 3V due to it’s difference with the cathode, and the charger has 0V at negative side (side connected to anode) while 5V at positive side, there is a pressure difference of 2 volt that will push current into the battery. Since positive is connected to positive and negative to negative, no electrons can flow from one device to the other, since cathode is already full of electrons and lithium in discharged state. In fact, the charger will put a high voltage on the cathode side, making the electrons in there high energy and pushing them out into the charger’s circuit, back into the anode. Electrons cannot enter a terminal having higher voltage than they do, and since the anode has now lower voltage and less electrons, the electrons naturally flow into that side again.
To have extra clarity, it must be pointed out that electrons don’t flow based on room availability, so to speak, but instead based on voltage difference, so based on pressure. If my charger has 5V and battery 3.7V, the electrons get pushed out of the cathode side even if that side isn’t full yet, making it so that I can charge even at 80% battery. So it’s all about relative voltage. The charger holds electrons at a lower potential and raises the battery cathode to the charger’s higher potential, which forces out electrons.
Something that’s actually quite interesting is, the charging rate occurs because of the voltage difference between the battery and the charger. If my battery is at 50% and has 3.6V going on, while at 0% it only has 3V going on, that means that at lower percentages of the battery, there’s a bigger voltage difference between the battery and the charger, making it charge faster in such a state than a nearly full state. This explains why charging from 90 to 100% takes so long. No such asymmetry exists while the battery is discharging.